Ammonia Solution Ammonium Hydroxide Chemical Structure and Storage Tips

1:A Closer Look
Ammonia solution ammonium hydroxide is an important chemical widely used in laboratories, industries, and cleaning applications. This compound forms when ammonia gas dissolves in water and creates a basic solution. Scientists and manufacturers value this solution because of its strong alkaline nature and its ability to react with many substances.
The chemical plays a major role in chemical synthesis, water treatment, agriculture, textile processing, and household products. Researchers also use it in analytical chemistry and laboratory experiments. Because the solution is reactive and can release ammonia vapors, proper understanding of its chemical structure and safe storage practices is essential.
This article tells the chemical structure of ammonium hydroxide, its preparation, practical applications, and the most effective storage methods. By understanding these aspects, laboratories, manufacturers, and users can handle the chemical safely and maintain its stability for long periods.
2:Overview of Ammonium Hydroxide
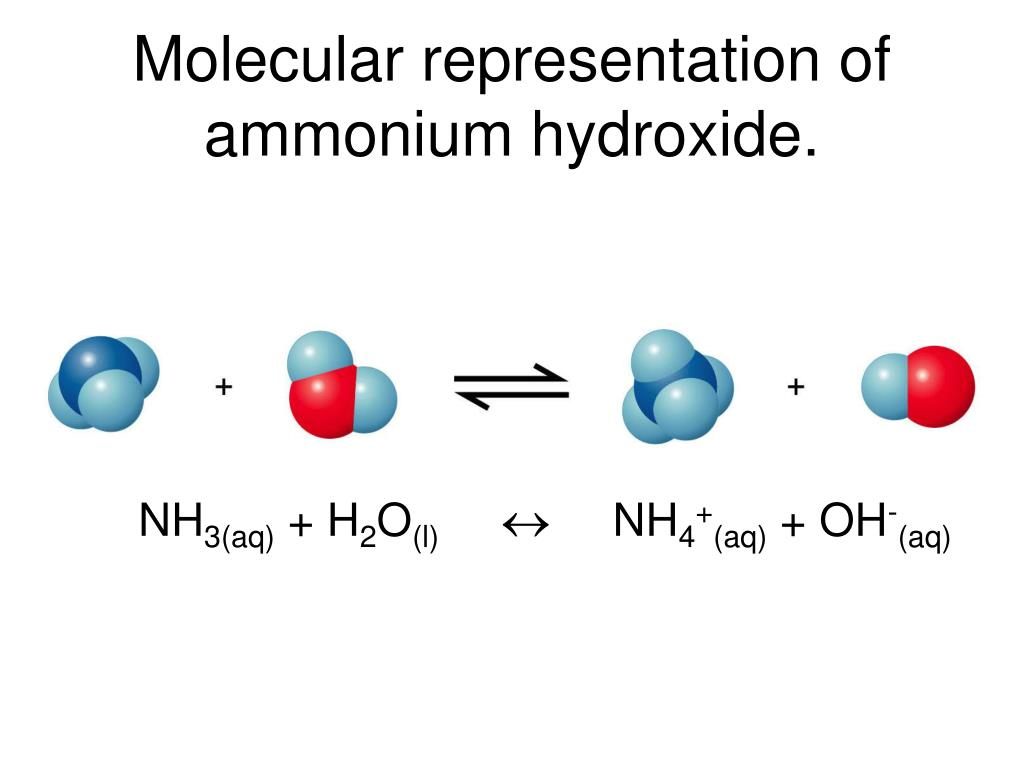
Ammonium hydroxide is not a solid compound that exists independently. Instead, it is a solution formed when ammonia dissolves in water. In this process, ammonia reacts partially with water molecules and produces ammonium ions and hydroxide ions.
Chemically, the reaction can be represented as:
NH₃ + H₂O ⇌ NH₄⁺ + OH⁻
This equilibrium reaction explains why the solution behaves as a weak base. The hydroxide ions increase the pH of the solution and make it alkaline. Because of this property, ammonium hydroxide is useful in neutralization reactions and many industrial processes.
The solution typically contains a mixture of dissolved ammonia, water, ammonium ions, and hydroxide ions. The concentration can vary depending on the amount of ammonia dissolved in water. Commercial solutions may range from low concentration to highly concentrated forms used in industrial applications.
3:Ammonium Hydroxide Formula
The ammonium hydroxide formula is commonly written as NH₄OH. Although this formula represents the composition of the solution, scientists understand that the compound actually exists in equilibrium between ammonia and water molecules rather than as a stable molecular compound.
This formula helps chemists describe the behavior of the solution in chemical reactions. It indicates the presence of ammonium ions and hydroxide ions, which are responsible for the alkaline properties of the solution.
Because of this structure, ammonium hydroxide can participate in acid–base reactions, precipitation reactions, and complex formation reactions. These characteristics make it a valuable reagent in both laboratory and industrial chemistry.
4:Molecular Structure of Ammonium Hydroxide
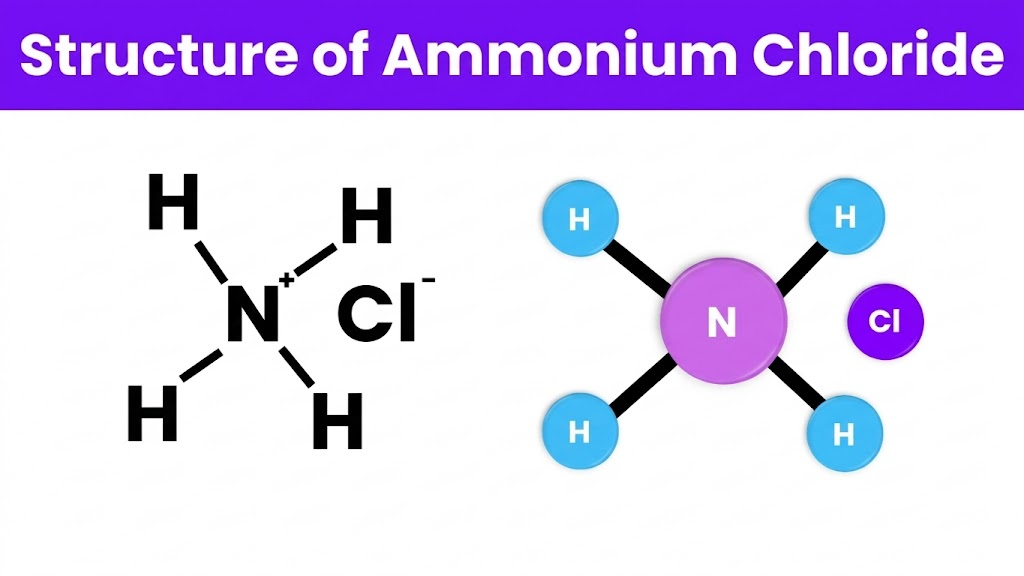
The chemical structure of ammonium hydroxide depends on the interaction between ammonia and water molecules. Ammonia contains one nitrogen atom bonded to three hydrogen atoms. The nitrogen atom also has a lone pair of electrons, which makes ammonia capable of accepting a proton.
When ammonia dissolves in water, it interacts with water molecules and forms ammonium ions (NH₄⁺). In this process, the nitrogen atom accepts a proton from water. At the same time, the water molecule loses a proton and becomes a hydroxide ion (OH⁻).
This process produces an equilibrium mixture that contains ammonia, ammonium ions, and hydroxide ions. Because the reaction is reversible, the solution constantly shifts between these forms depending on temperature and concentration.
The presence of hydroxide ions is responsible for the alkaline nature of the solution. These ions increase the pH and allow the chemical to neutralize acids, remove grease, and participate in chemical synthesis.
5:Industrial and Laboratory Preparation Method
The ammonia solution ammonium hydroxide preparation process usually involves dissolving ammonia gas in water under controlled conditions. Manufacturers pass ammonia gas through purified water while maintaining proper temperature and pressure.
During this process, ammonia dissolves rapidly and forms the ammonium hydroxide solution. The concentration of the final product depends on the amount of ammonia gas absorbed by the water.
Industrial facilities often use special absorption towers to produce large quantities of the solution. These towers allow efficient mixing of ammonia gas and water while preventing the escape of ammonia vapors.
Key point to know In laboratory environments, chemists can prepare smaller quantities by carefully bubbling ammonia gas into distilled water. Because ammonia gas has a strong odor and can irritate the respiratory system, laboratories must conduct this process inside a fume hood with proper ventilation.
6:Physical and Chemical Properties
Ammonium hydroxide solution has several distinct physical and chemical properties that make it useful in different applications.
The solution appears as a clear and colorless liquid with a strong and pungent ammonia smell. It is highly soluble in water and forms a basic solution with a high pH.
One important property of the compound is its volatility. Ammonia can easily escape from the solution, especially at higher temperatures. For this reason, containers must remain tightly sealed during storage.
The solution also reacts with acids to form ammonium salts. These reactions make it useful in neutralization processes and chemical synthesis.
Another important characteristic is its ability to form complexes with metal ions. This property allows chemists to use the solution in analytical chemistry to detect certain metals.
7:Ammonium Hydroxide Uses

Ammonium hydroxide uses cover a wide range of industrial, commercial, and laboratory applications.
In laboratories, chemists use it as a reagent for qualitative analysis. It helps identify metal ions through precipitation reactions and complex formation.
Industries use the solution in the production of fertilizers, plastics, dyes, and pharmaceuticals. It also plays a role in manufacturing cleaning agents and disinfectants.
Another important application involves water treatment. The chemical helps adjust pH levels and remove impurities from water systems.
In the textile industry, manufacturers use it during fabric processing and dyeing. It assists in controlling acidity and improving the quality of finished materials.
Household cleaning products also contain diluted forms of the solution. Its alkaline nature helps break down grease, oils, and stains on surfaces.
8:Role in Laboratory Chemistry

Ammonium hydroxide is one of the most commonly used reagents in analytical laboratories. Chemists use it to control pH during experiments and to produce precipitates that help identify different metal ions.
For example, the solution can react with metal salts to form insoluble metal hydroxides. These reactions allow chemists to determine the presence of metals such as copper, iron, or zinc in a sample.
The chemical also helps prepare buffer solutions that maintain stable pH conditions during experiments. These buffers are important in biological and chemical research.
Because of its versatility, ammonium hydroxide remains an essential reagent in many laboratory techniques.
9:Storage and Handling Tips

Proper storage is essential for maintaining the stability and safety of ammonium hydroxide solution. Because the solution can release ammonia vapors, containers must remain tightly sealed.
Chemicals should be stored in corrosion-resistant containers made from materials such as polyethylene or glass. These materials prevent chemical reactions between the solution and the container.
The storage area should remain cool, dry, and well ventilated. High temperatures can increase the evaporation of ammonia gas and reduce the concentration of the solution.
Direct sunlight should also be avoided because heat can accelerate chemical changes and vapor release.
Key point to know: Safety equipment such as gloves, goggles, and protective clothing should always be used when handling the chemical. Adequate ventilation prevents inhalation of ammonia vapors, which can irritate the respiratory system.
10:Transportation and Packaging

Transporting ammonium hydroxide requires careful packaging and labeling. Containers must remain tightly closed to prevent leakage and vapor release.
Regulatory agencies often classify the solution as a hazardous chemical because of its corrosive nature and strong fumes. For this reason, transportation guidelines require proper hazard labels and documentation.
Industrial suppliers usually ship the solution in sealed plastic drums, chemical tanks, or specialized containers designed for corrosive liquids.
Safe transport ensures that the chemical reaches laboratories or manufacturing facilities without contamination or loss of concentration.
11:Ammonium Hydroxide Solution in Industrial Processes

The ammonium hydroxide solution plays an important role in several industrial processes. Manufacturers use it to control pH levels during chemical reactions and to produce various compounds.
One major application occurs in fertilizer production. The chemical contributes to nitrogen-based fertilizers that support plant growth.
Another industrial use involves the production of rubber and latex products. The solution helps stabilize latex and prevent unwanted coagulation during processing.
Food processing industries sometimes use controlled amounts of ammonium hydroxide to adjust acidity levels in certain products.
In addition, manufacturers rely on the solution for metal cleaning, surface treatment, and chemical synthesis.
12:Reliable Sources to Buy This Chemical

Many chemical suppliers and laboratory equipment companies provide this compound for research and industrial use. When searching where to buy ammonia solution ammonium hydroxide, buyers should choose reliable chemical distributors that follow safety and quality standards.
Reputable suppliers provide high-purity chemicals with clear labeling, safety documentation, and certificates of analysis. These documents confirm the concentration and purity of the chemical.
Online chemical marketplaces, laboratory supply stores, and industrial chemical manufacturers often distribute ammonium hydroxide in different concentrations depending on the intended use.
Before purchasing, buyers should verify regulatory requirements and ensure that storage and handling facilities meet safety guidelines.
13:Availability of This Chemical in the Market

Many chemical distributors offer ammonia solution ammonium hydroxide for sale in multiple concentrations and packaging sizes. These may include small laboratory bottles, medium-sized containers for research facilities, or large industrial tanks for manufacturing plants.
Suppliers usually provide detailed safety information, product specifications, and recommended storage conditions. Buyers should review these details carefully before purchasing the chemical.
Selecting the correct concentration is also important. Laboratories often use lower concentrations, while industrial applications may require stronger solutions.
Reliable suppliers ensure consistent quality and safe packaging to protect the chemical during storage and transportation.
14:Safety Considerations
Ammonium hydroxide is a useful chemical, but improper handling can cause health risks. The solution is corrosive and may irritate the skin, eyes, and respiratory system.
Direct contact with the liquid can cause burns or irritation, so protective equipment is essential when handling it. Goggles, gloves, and protective clothing help prevent exposure.
Inhalation of ammonia vapors may cause coughing, breathing discomfort, or irritation in the nose and throat. Proper ventilation or working inside a fume hood reduces this risk.
Spills should be cleaned immediately using appropriate safety procedures. Dilution with water and neutralization with mild acids can help manage small spills under controlled conditions.
Following safety guidelines ensures that the chemical remains useful without posing unnecessary risks.
15:Environmental Impact

Like many industrial chemicals, ammonium hydroxide can affect the environment if released in large amounts. High concentrations may alter the pH of water bodies and harm aquatic life.
Industrial facilities must follow environmental regulations when disposing of waste solutions. Proper neutralization and treatment prevent contamination of water systems.
Responsible handling, storage, and disposal practices reduce environmental risks while allowing industries to benefit from the chemical.
For more information also read this: https://westrnhealth.com/chemical/how-to-use-and-store-glacial-acetic-acid-safely/
Bottom line
Ammonia solution ammonium hydroxide is an important alkaline chemical widely used in laboratories, industries, and cleaning products. The solution forms when ammonia gas dissolves in water and creates an equilibrium mixture of ammonia, ammonium ions, and hydroxide ions. This structure gives the chemical its basic properties and makes it useful in many reactions.
Industries rely on ammonium hydroxide for fertilizer production, textile processing, chemical synthesis, and water treatment. Laboratories use it in analytical chemistry and pH control. Because the chemical can release ammonia vapors and cause irritation, proper storage and safety measures are essential.
Keeping the solution in tightly sealed containers, storing it in cool and ventilated areas, and using protective equipment helps maintain safety and stability. When handled responsibly, ammonium hydroxide remains a valuable chemical for research, manufacturing, and industrial processes.
Helpful Questions Readers Often Ask
1. What is ammonium hydroxide used for?
Ammonium hydroxide is commonly used in laboratories, cleaning products, fertilizer production, textile processing, and water treatment.
2. Is ammonium hydroxide the same as ammonia?
No. Ammonia is a gas (NH₃), while ammonium hydroxide is the aqueous solution formed when ammonia dissolves in water.
3. Why is ammonium hydroxide considered a weak base?
It is called a weak base because only a small portion of dissolved ammonia reacts with water to form hydroxide ions.
4. How should ammonium hydroxide be stored safely?
It should be stored in tightly sealed containers, in a cool and well-ventilated area, away from heat and direct sunlight.
5. Is ammonium hydroxide dangerous to handle?
It can be corrosive and produce irritating vapors, so users must wear gloves, goggles, and work in properly ventilated areas.








